Introduction
Celiac disease is an inborn hypersensitivity to gluten, to be more precise, a hypersensitivity to the sub-fraction of gluten, called “gliadin”. Alternative names for this condition are: sprue, celiac sprue and gluten enteropathy.
Recently a new test has been developed, which is very specific and sensitive, called endomysial antibody (EMA) titre. Most noteworthy, this test demonstrated that many more people have celiac sprue than it was previously assumed.
For instance, in Ref. 9 the authors found by studying a sample population in New Zealand that 1.2% of the population were positive for celiac disease, which was 3-fold more common than previously thought. A recent review cited that one in 133 people in North America have celiac disease (Ref. 34).
Pathophysiology of celiac’s disease
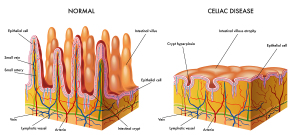
The pathophysiology of celiac disease (CD) is such that the antibodies against gliadin (from wheat , rye, barley or other food products) form immune complexes when they get into contact with gliadin in the small intestine. Immunofluorescence visualizes these IgA deposits in small bowel biopsy samples or skin biopsy samples. This leads to chronic inflammation, scarring and atrophy (three different degrees of it shown). The end result is malabsorption. This means that the small intestine can no longer absorb the normal amount of nutrients as one would expect to normally occur. This malabsorption syndrome is what causes all of the symptoms of this gluten intolerance.
HLA markers
According to Ref.34, 90% to 95% of CD patients carry a genetic marker HLA-DQ2, which can be tested for in the lab. The remainder of CD patients are HLA-DQ8 positive. However, 10% to 30% of the general population without symptoms of CD have a positive HLA-DQ2 test. This subpopulation is likely at risk of developing CD down the road and needs to be screened more often with blood tests to see whether the titers are changing. If there is any doubt a small bowel endoscopy with biopsy needs to be performed and the samples are examined for IgA deposits by immunofluorescence.
Using these screening tools it has become apparent that most cases of CD present with non-gastrointestinal symptoms, such as itchy skin (dermatitis herpetiformis), osteoporosis or osteopenia, delayed puberty, short stature, iron deficiency, hepatitis or joint pains.
Signs and symptoms
Often and perhaps even most of the time celiac disease has no symptoms, at least not initially. The more severe cases would have some fatty stools and bowel movements that are more frequent. In the past it was thought that this was solely a disease of toddlers who would present with a failure to grow, have abdominal cramps and diarrhea. As a result we now know that the gastrointestinal symptom presentation is only the tip of the iceberg of CD symptoms. 90% of CD patients have no gastrointestinal symptoms. The older child and adults may present with symptoms of dermatitis herpetiformis, dental enamel defects, osteoporosis in males with bone and muscle aches. It seems like iron deficiency is very common as is an elevation of transaminases from a non-specific reactive hepatitis or an autoimmune hepatitis. Consequently, all of these symptoms can disappear when a strict gluten free diet is followed.
Gastrointestinal symptoms
In those children or adults who have gastrointestinal symptoms there may be some abdominal cramps after certain foods. When these patients had celiac disease as children already, body growth is often inhibited and for that reason these patients often are of short stature. Other symptoms can be an itchy skin condition with skin lesions distributed in a symmetric fashion over the knees, elbows, buttocks and the back. This is due to dermatitis herpetiformis. We now know that this skin disease is merely another presentation of celiac disease and the EMA titre is often positive in these patients as well. As a result, other consequences of the malabsorption, such as iron deficiency, can lead to anemia (microcytic anemia, often more in children).
Other symptoms
First of all, vitamin B12 and folic acid deficiency lead to another form of anemia, which will look different under the microscope (megaloblastic anemia, often in adult celiac disease). Also, the lack of vitamin D can lead to bone deformities and rickets. There might be bone pain as well. Furthermore, the more atrophy in the small intestine there is, the less absorption of sugars such as xylose will take place and this leads to an osmotically driven diarrhea. Along with the diarrhea there is a loss of valuable minerals and protein. The end result is a slow form of starvation. Finally, in women there is a problem with fertility and lack of menstrual periods. Late onset of puberty can be another sign of CD.
Joint aches and pains
Some children present with joint aches and pains, which can develop into chronic arthritis. As arthritis is typically more a disease of older people, symptoms of chronic arthritis in children should therefore make the physician very suspicious of CD.
Likewise, in some people dental enamel defects of the teeth may be the only signs of celiac disease as shown in this blog.
Diagnosis
Because the symptoms can be so subtle, it was difficult in the past to make the diagnosis. Now there is the more specific endomysial antibody titre test as mentioned above, which will clearly show whether or not the patient has celiac disease. Another test measures antibodies against tissue transglutaminase (TTG IgA). This latter test is highly sensitive and specific for CD. However, the physician needs to think about ordering this test.
Other tests
Other tests that might be useful: if there is a combination of low calcium, potassium and sodium, coupled with a low albumin count and a high alkaline phosphatase, this should be a trigger to order the EMA titre or TTG IgA test. To determine the degree of malabsorption the 5 gram D-xylose test can be ordered, which will determine what percentage of this sugar is absorbed. The most direct test is an endoscopic procedure where the gastroenterologist visualizes the first part of the small bowel (jejunum) and takes several mini biopsies. These will confirm the presence of celiac disease and also the severity and the amount of atrophy. This has some prognostic implications, as not every case will respond to a simple gluten free diet. The gold standard to make the definitive diagnosis of CD is still a small bowel biopsy with immunofluorescence analysis to show IgA deposits.
Treatment
Before treatment is instituted, the diagnosis must have been established beyond a shadow of a doubt (see above). The main part of the therapy is to strictly avoid gluten in the diet. This is a major step in anybody’s life and unfortunately, there is no exception and no holiday from this for the rest of the life of the person with celiac disease. On the other hand, once the patient is used to a strict gluten free diet, being symptom free is so rewarding that most patients have no problem staying on this diet lifelong. Dr. Sigman stated in Ref. 34 “It is extremely important that physicians take the time to explain the importance of remaining on the gluten-free diet for life”.
Follow-up visits
Daily multivitamins are also recommended as gluten-free pastas and flours lack B vitamins, folic acid and iron. Ref. 34 recommends that a yearly visit to the treating physician should include ordering tests such as TTG IgA , iron tests, complete blood count, albumin, vitamin B12 and folic acid, thyroid function tests and other micronutrient levels depending on the clinical situation. The gluten-free diet has been shown to reduce the risk of micronutrient deficiencies. It will possibly also diminish the risk of developing other immune disorders and intestinal lymphoma.
Celiac disease and gluten free diet
I would like to explain why it is so important to take this dietary step seriously.
Let me explain this by way of an analogy to an asthmatic patient who has been diagnosed with an allergy to cat and dog hair. The allergist says that unfortunately there is no allergy shot that can be given, but the patient must avoid indoor exposure to cats and dogs. If the patient abides by this recommendation, he/she will be fine with the asthma. However, if the asthmatic has a cat that he/she really loves and does not get rid of and wants to keep inside the house, there will likely be a serious asthma attack down the road due to dander accumulation in the house, which sensitizes the patient.
Damage from circulating antibodies
Alternatively, there could be a slow process of closing down the airways from the immune complex (cat dander firmly attached to the circulating antibodies in the asthmatic’s system), which would then lead to lung fibrosis, a dangerous irreversible lung condition similar to end stage emphysema.
The same phenomenon of circulating antibodies that have to be taken seriously, is happening in celiac disease, except that the antigen is not inhaled, but enters the system through the gut from the food we eat. The target organ here are not the lungs, but the lining of the small intestine (jejunum). With the asthmatic the transport of air across the lung is at stake. With celiac disease the transport of all of the nutrients through the gut wall is at stake (malabsorption develops).
Seeing a dietician
Because the gluten free diet is so crucial, a referral to a knowledgeable dietitian is important. There is a need for supplements for a period of time. In more serious cases supplementation may have to be given on an ongoing basis. Your doctor will advise you regarding your particular case.
From time to time the physician will want to repeat the EMA titre or tissue transglutaminase (TTG IgA). The authors of Ref. 10 have shown that the EMA titre can successfully monitor gluten diet compliance. The titre disappears in a compliant patient, but reoccurs in non compliant patients. A similar observation, although more crude, is the xylose absorption test, which will be normal with a compliant patient, but return to abnormal values with non-compliance.
The gluten-free diet will prevent long-term complications from uncontrolled intestinal inflammation.
Practical point and prevention
__________________________________________________________
Hummel et al. (Ref. 11) found an interesting connection between diabetes and celiac disease. This group followed children of patients with type I diabetes (insulin dependent) and checked them for EMA titers. To their surprise they found that there is a significantly higher percentage of children with celiac disease when compared to the normal population. Also, a significant number of these children, who were not yet symptomatic, showed established celiac disease on endoscopic biopsy. It appears that with more experience targeted screening might be possible for high risk groups of celiac disease using the endomysial antibody (EMA) titre or blood tests for tissue transglutaminase (TTG IgA).
__________________________________________________________
References
1. M Frevel Aliment Pharmacol Ther 2000 Sep (9): 1151-1157.
2. M Candelli et al. Panminerva Med 2000 Mar 42(1): 55-59.
3. LA Thomas et al. Gastroenterology 2000 Sep 119(3): 806-815.
4. R Tritapepe et al. Panminerva Med 1999 Sep 41(3): 243-246.
5. The Merck Manual, 7th edition, by M. H. Beers et al., Whitehouse Station, N.J., 1999. Chapters 20,23, 26.
6. EJ Simchuk et al. Am J Surg 2000 May 179(5):352-355.
7. G Uomo et al. Ann Ital Chir 2000 Jan/Feb 71(1): 17-21.
8. PG Lankisch et al. Int J Pancreatol 1999 Dec 26(3): 131-136.
9. HB Cook et al. J Gastroenterol Hepatol 2000 Sep 15(9): 1032-1036.
10. W Dickey et al. Am J Gastroenterol 2000 March 95(3): 712-714.
11. M Hummel et al. Diabetologia 2000 Aug 43(8): 1005-1011.
12. DG Bowen et al. Dig Dis Sci 2000 Sep 45(9):1810-1813.
13. The Merck Manual, 7th edition, by M. H. Beers et al., Whitehouse Station, N.J., 1999.Chapter 31, page 311.
14. O Punyabati et al. Indian J Gastroenterol 2000 Jul/Sep 19(3):122-125.
15. S Blomhoff et al. Dig Dis Sci 2000 Jun 45(6): 1160-1165.
16. M Camilleri et al. J Am Geriatr Soc 2000 Sep 48(9):1142-1150.
17. MJ Smith et al. J R Coll Physicians Lond 2000 Sep/Oct 34(5): 448-451.
More references:
18. YA Saito et al. Am J Gastroenterol 2000 Oct 95(10): 2816-2824.
19. M Camilleri Am J Med 1999 Nov 107(5A): 27S-32S.
20. CM Prather et al. Gastroenterology 2000 Mar 118(3): 463-468.
21. MJ Farthing : Baillieres Best Pract Res Clin Gastroenterol 1999 Oct 13(3): 461-471.
22. D Heresbach et al. Eur Cytokine Netw 1999 Mar 10(1): 7-15.
23. BE Sands et al. Gastroenterology 1999 Jul 117(1):58-64.
24. B Greenwood-Van Meerveld et al.Lab invest 2000 Aug 80(8):1269-1280.
25. GR Hill et al. Blood 2000 May 1;95(9): 2754-2759.
26. RB Stein et al. Drug Saf 2000 Nov 23(5):429-448.
27. JM Wagner et al. JAMA 1996 Nov 20;276 (19): 1589-1594.
28. James Chin, M.D. Control of Communicable Diseases Manual. 17th ed., American Public Health Association, 2000.
29. The Merck Manual, 7th edition, by M. H. Beers et al., Whitehouse Station, N.J., 1999. Chapter 157, page1181.
30. Textbook of Primary Care Medicine, 3rd ed., Copyright © 2001 Mosby, Inc., pages 976-983: “Chapter 107 – Acute Abdomen and Common Surgical Abdominal Problems”.
31. Marx: Rosen’s Emergency Medicine: Concepts and Clinical Practice, 5th ed., Copyright © 2002 Mosby, Inc. , p. 185:”Abdominal pain”.
32. Feldman: Sleisenger & Fordtran’s Gastrointestinal and Liver Disease, 7th ed., Copyright © 2002 Elsevier, p. 71: “Chapter 4 – Abdominal Pain, Including the Acute Abdomen”.
33. Ferri: Ferri’s Clinical Advisor: Instant Diagnosis and Treatment, 2004 ed., Copyright © 2004 Mosby, Inc.
34. Terry Sigman, assistant professor in the Division of Gastroenterology at McGill University and at Montreal Children’s Hospital: The Medical Post Vol.47, No.6, April 5, 2011, page 65 “So you think you know celiac disease?”