Introduction
There are three major disease entities that make up chronic leukemia:
- Chronic lymphocytic leukemia (CLL)
- Chronic myelogenous leukemia (CML)
- Myelodysplastic syndrome (MDS)
Some facts about bone marrow
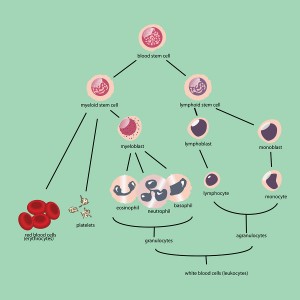
The soft tissue inside the long bones, the pelvic bones, the vertebral bodies and the breast bone (sternum) is called bone marrow. This is the tissue, which regenerates all the blood cells and there is a finely tuned balance between the circulating blood cells and the reservoir of the bone marrow. It is important to recognize that all of the blood cells originate from stem cells, which are immature cells distributed throughout the bone marrow . All of the other blood cells are derived from these stem cells. The lymphocytes are the immune cells of the body. They fight infection in two ways: through the antibody producing B cells and through the T cells. The B cells are lymphocytes producing antibodies against bacteria and viruses. The T cells, which are lymphocytes that matured with the help of the thymus gland, fights cancer cells, parasites and transplants that are mismatched. Both B cells and the T cells came from the same stem cell. Granulocytes are the major bulk of the white blood cells and they are the major component of pus cells that will combat bacteria and are involved in wound healing. They are formed under the influence of the “colony stimulating factor”, a kind of tissue hormone, which programs the stem cells to differentiate into granulocytes. The hormone erythropoietin from the kidney will stimulate stem cells to develop into a red blood cell. And finally, the platelets are derived from the stem cell via a precursor cell called megakaryocyte. In the following outline we are dealing with cancers of the lymphocyte origin ( CLL) and the granulocyte origin( CML). The MDS involves various precursor cells of the bone marrow (Ref. 1 and 2).
General remarks regarding chronic leukemia
CLL occurs mostly in older people (aged more than 60 years) and is twice as frequent in men than in women. CLL is rare in Japan and China. It can be found more frequently in some families and when one family member is diagnosed with it, other first-degree family members have a threefold risk for CLL or another blood disorder. CLL has an age related risk of occurrence: between the age of 35 and 59 there is an incidence of 5 per 100, 000 persons in the US, but at age 80 to 84 this increases to 30 per 100,000 persons, a 6-fold increase. No link of radiation has been established with CLL, nor is there any known leukemia virus that would cause it. CML occurs in either sex and is a leukemia of middle age (most common around age 45 years). It is uncommon before 10 years of age. In this leukemia 95% of patients have a chromosome marker at the chromosome 22 (called “Philadelphia chromosome”), which is characteristic for this type of leukemia. A chromosomal translocation at this chromosome is believed to be the cause of CML. This occurs at the stem cell level or the myeloblast level (see image above). From this point on it takes an average of 4 years before a blast crisis occurs. At that point more than 30% of the normal bone marrow cells have been replaced by blast cells which have become a huge cell clone; this puts pressure on the precursors of red blood cells causing severe anemia and also interferes with normal platelet production resulting in low platelet counts at the same time. This can result in severe bleeding (due to low platelet counts), weakness as well as proneness to infections (low red blood and immune cell counts). At that critical blast crisis hundreds of thousand of blast cells per cubic millimeters will travel in the blood stream and invade vital organs such as the brain, kidneys, lungs and liver. The mean median survival time after blast crisis is only about 2 months. It can be extended to about 10 months, if remission can be achieved. With the introduction of interferon therapy we have a mechanism to delay or prevent the development of the blast crisis. Survival rates have increased from about 3.5 years before interferon therapy to about 7 years. However, early diagnosis is necessary. Despite best efforts this was the best until spring of 2001 that medicine could offer. But as pointed out below, with the new tyrosine kinase inhibitors, such as Gleevec, an exciting possibility has opened up to cure this terrible disease (see below).
Gleevec – The “New Kid On The Block” To Treat CML
_______________________________________________________
Genetically based therapies are the only hope for a longterm solution. New research is starting to come out regarding this. A case in point is the new medicine, which was called STI 571 and has been released in May 2001 by the FDA under the trade name of “Gleevec”. It is a tyrosine kinase inhibitor. Molecular biological research has shown in the past that CML cells have a modified gene, which activates the production of tyrosine kinase that in turn is responsible for the rapid cell division. It follows from this that a medication that would inhibit this enzyme should stop CLL cells from growing. The beauty of this line of research is that everything worked as planned and the human studies to show its safety and effectiveness have been done (Ref.3 and 4). The data show that this new medication is safe, has few side-effects and that it also is effective against CML with 53 out of 54 patients having shown a complete remission (Ref. 3).
_______________________________________________________
References:
1. Cancer: Principles &Practice of Oncology. 4th edition. Edited by Vincent T. DeVita, Jr. et al. Lippincott, Philadelphia,PA, 1993. Chapter on Chronic Leukemia.
2. Cancer: Principles&Practice of Oncology. 5th edition, volume 2. Edited by Vincent T. DeVita, Jr. et al. Lippincott-Raven Publ., Philadelphia,PA, 1997. Chapter on Chronic Leukemia.
3. BJ Druker et al. N Engl J Med 2001 Apr 5;344(14):1031-1037.
4. MJ Mauro et al. Curr Oncol Rep 2001 May;3(3):223-227.
5. Conn’s Current Therapy 2004, 56th ed., Copyright © 2004 Elsevier
6. Ferri: Ferri’s Clinical Advisor: Instant Diagnosis and Treatment, 2004 ed., Copyright © 2004 Mosby, Inc